|
There was no history suggestive of collodion membrane. The birth weight of the baby was 2.3 kg and the length of the baby was 36 cm. There was a history of consanguineous marriage and prolonged labor and baby was delivered by normal vaginal delivery. The rare disease awareness ribbon is a zebra-striped ribbon.A one-and-half year old boy presented with generalized scaling over whole body and redness over both cheeks. The awareness day was originally started by the European Organization for Rare Diseases and is now recognized globally. Rare Disease Awareness Day is held globally on February 29 - February 28 on non-leap years. The symbol for rare disease awareness is a zebra-striped ribbon. 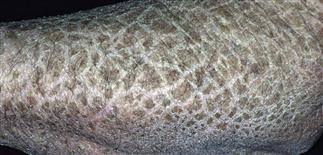
Japan: In Japan, the legal definition of a rare disease, JPN Pharmaceutical Orphan Drug Law (Oct 1993), is one that affects fewer than 50,000 patients in Japan, or about 1 in 2,500 people.The EU Orphan Regulation No 141/2000 (Dec 1999), defines an rare disease as a condition with a prevalence in the EU of 5 in 10,000 people (or fewer). European Union (EU): The EU defines rare diseases as conditions that affect less than 1 in 2,000 people, but many are much rarer.That represents approximately 3 million Canadians and their families facing a debilitating disease that severely impacts their lives, making finding treatments for them a serious public health concern for the country. Canada: According to the Canadian Organization for Rare Diseases (CORD), rare diseases affect one in 12 Canadians (two-thirds of whom are children).While individual diseases may be rare, the total number of people with a rare disease is large. The total number of Americans living with a rare disease is currently estimated at between 25-30 million. Before the passage of rare disease laws in the United States, patients diagnosed with a rare disease were denied access to effective medicines because prescription drug manufacturers rarely could make a profit from marketing drugs to such small groups. This definition was created by United States Congress in the Orphan Drug Act (ODA) of 1983. United States: In the United States (US), a rare disease is defined as one that affects fewer than 200,000 people.Rare disease definitions used in medical literature and by national health plans are divided, with definitions ranging from 1 in 1,000 people to 1 in 200,000 people. Some definitions are based on the number of people living with a disease, and other definitions include factors, such as existence of adequate treatment(s) or the severity of the disease. There is no single, widely accepted definition for rare diseases. On average 1 in 17 people will be affected by a rare disease at some point during their lives. Global Genes estimates over 300 million people worldwide are living with a rare disease. Rare diseases include, some very rare infectious diseases, rare forms of autoimmune disorders, and rare cancers. There are approximately 7,000 rare diseases (RD). 
Most genetic disorders are rare diseases, but not all rare diseases are caused by genetic factors. The term orphan disease is sometimes used as a synonym for rare disease. In the United States the Congress in the Orphan Drug Act (ODA) of 1983 defines a rare disease as one that affects fewer than 200,000 people.Īdvertisements Main Digest Defining Rare Diseases The European Union (EU) defines rare diseases as conditions that affect less than 1 in 2,000 people, but many are much rarer. Synopsis: Information includes a list of rare diseases and the definition of the term orphan disease, which is sometimes used as a synonym for rare disease. Author: Disabled World | Contact: Disabled World ()
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
